commentary Commentary
Commentary: Without a vaccine, how can pregnant mothers protect themselves against COVID-19?
There are trials going on in other countries for pregnant women to be vaccinated against COVID-19. If this is approved, what are the risks for an expectant mother to get a shot and without it, can they still be safe? Professor David Matchar of Duke-NUS Medical school explains.
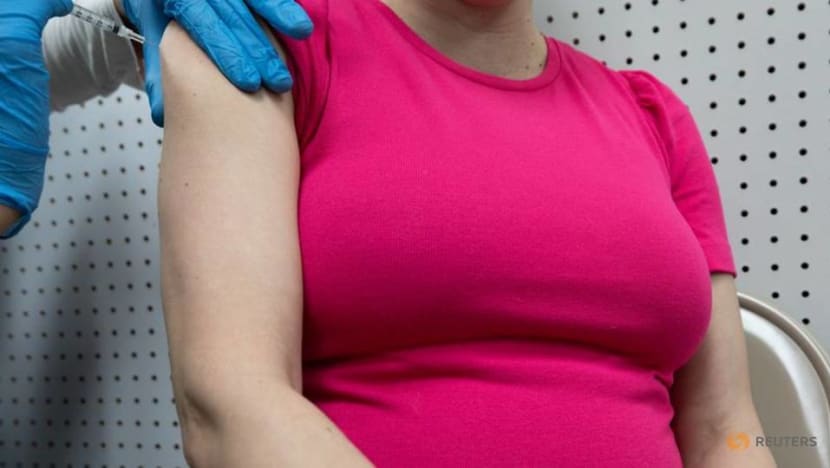
A pregnant woman receives a vaccine for the coronavirus disease (COVID-19) at Skippack Pharmacy in Schwenksville, Pennsylvania, on Feb 11, 2021. REUTERS/Hannah Beier)
SINGAPORE: The nine months of a pregnancy are often a period of great joy, anticipation and a fair share of trepidation. On top of all the worries and anxieties about becoming new parents, welcoming another life into the family, we also worry about the health of the mother and foetus.
The pandemic has only heightened these worries, especially given that pregnant women are at higher risk of severe COVID-19 than non-pregnant women, and COVID-19 has been associated with an increased risk of pre-term birth, according to the World Health Organization.
Getting vaccinated, therefore, is a priority for many. But while vaccination remains the best defence against COVID-19, the immediate benefit of vaccination depends on the local COVID-19 situation.
READ: Commentary: Concerns over long-term side effects could hold back Singapore’s COVID-19 vaccination programme
In regions experiencing high rates of infection, vaccination may seem a favourable choice as your risk of contracting COVID-19 is much greater.
For Singapore, where current local transmission of the virus is extremely low, the risk of a pregnant woman contracting the virus is similarly low and hence the immediate benefit from vaccination is less significant.
However, if the risk of a surge in community transmissions here increases sharply, the likely policy would be to rapidly move to vaccinate pregnant women, among other measures.
READ: The fears and resilience of having a baby in the time of COVID-19
WHY PREGNANT WOMEN ARE NOT IN LINE FOR VACCINATION
For context, clinical trials as a rule exclude some groups of people including pregnant women unless they are the specific subject of the study.
As pregnant women were not included in the trials that evaluated the safety and efficacy of the new COVID-19 vaccines, health authorities around the world remain cautious and are recommending that they defer vaccination until more data becomes available.
And such trials are now underway. Pfizer and BioNTech launched the first COVID-19 vaccine trial for pregnant women in February 2021 to evaluate the safety and effectiveness of their COVID-19 vaccine in healthy pregnant women.
At the same time, international recommendations – and those from the Ministry of Health (MOH) in Singapore – are tempering exclusions as more clinical data of COVID-19 vaccines become available.
For instance, MOH has just relaxed its COVID-19 vaccination guidelines for some people with allergies because emerging scientific evidence has shown that these individuals can be vaccinated with a messenger ribonucleic acid (mRNA)-based COVID-19 vaccine as long as there are no other life-threatening signs and symptoms of anaphylaxis.
Women who are breastfeeding can now also go for the vaccine if they are willing to stop breastfeeding for five to seven days after being vaccinated as a cautionary measure.
READ: Commentary: Confinement without help during COVID-19 – Mums, you're stronger than you know
Likewise, when we have more data to conclude that COVID-19 vaccines are safe in pregnant women, we are likely to see a policy change in countries including in Singapore.
But the questions would then be: Should I take the vaccine? Will I experience the advantages with none of the disadvantages? To some people, the decision seems complicated given limited data about what might happen in the long term.
If you are on (or near) the fence on whether to get vaccinated, it may help to consider two things: There is no such thing as zero risk, and the best you can do is to decide by understanding and balancing the risks and benefits.
READ: Commentary: Here’s why taking the vaccine is necessary even if it’s optional
NO SUCH THING AS ZERO RISK
Scientifically speaking, pregnant women should not receive live-virus vaccines which may present a hypothetical risk to the foetus. One example is the Measles, Mumps, Rubella (MMR) vaccine which is a mixture of live but weakened viruses and not given during pregnancy.
Neither the Pfizer-BioNTech nor Moderna vaccine contains a live virus.
But no amount of research can guarantee absolute safety. What studies can do is to increase the confidence about the level of risk. Is it more like the risk of walking with green man at the intersection or jaywalking across the highway?

If you are pregnant, you may be troubled that the data on risk of vaccination of pregnant women is much less compelling than that for other adults.
However, with no evident signal of harm from more than 51,000 pregnant women participating in the v-safe Pregnancy Registry launched by US’ Center for Disease Control and Prevention, it gives us high confidence that the vaccine will do no immediate harm.
We should also consider a second outcome: Severe COVID-19 infections which are much more serious than a bad cold, with symptoms including life-threatening pneumonia, damage to multiple organs such as the heart, brain or kidneys, and even prolonged complications lasting years.
If pregnant women develop these severe symptoms, studies suggest that they are more likely to need intensive care than non-pregnant women.
READ: Commentary: Concerns over severe allergic reactions to COVID-19 vaccines don't outweigh benefits of vaccination
READ: Commentary: If mums are amazing, why do some workplaces discriminate against pregnant women?
BALANCING RISKS AND BENEFITS
The critical question to ask is, therefore, what the balance between the risk (major side effect of the vaccine) and benefit (avoidance of severe COVID-19) is.
Based on currently accumulated data released in a CDC weekly report, of the more than 4 million people who have received the first dose of Moderna's COVID-19 vaccine, 0.03 per cent experienced adverse events. The chances of having severe adverse symptoms is under 0.003 per cent.
The benefit can also be higher if a surge of infections is active or is likely in the future.
Risk estimates from Mathematica show that in the midst of a three-month COVID-19 surge, a pregnant woman has a greater than 1 per cent chance of both becoming infected and requiring hospitalisation. Effective vaccination has the benefit of avoiding the majority of those severe infections.
Hence if you live in a region experiencing high rates of infection, or in a country that is ripe for a surge due to low levels of population immunity, vaccination seems a favourable choice. For example, at least 20 states in the US provide vaccination for pregnant women. The same policy applies in Britain and Israel as well.
One additional benefit of vaccinating expectant mothers is the passing of protection to their babies, just like the flu shot or the tetanus, diphtheria and whooping cough vaccine (Tdap) which are actively recommended during pregnancy.
While further data is needed to confirm the maternal and neonatal protection from SARS-CoV-2 infection arising from vaccinations during pregnancy, a recent study in Israel of 20 women who received both doses of the Pfizer-BioNTech vaccine during their third trimester of pregnancy shows that antibodies were detected in their newborns through placental transfer.
Presuming vaccine studies confirm that vaccine is safe broadly, policy constraints for vaccination will decrease and more people will feel more confident with the choice to be vaccinated.
For those of you with a newfound choice, considering the possible consequences of the decision and the likelihood of those consequences allows some peace in the knowledge that whatever the outcome, you made the most well-informed decision you could.
In the meantime, as long as you “can’t” be vaccinated as a matter of public policy, or the result of a personal choice, there is one option – the same as before vaccines started to roll out and the one that will persist for everyone until the Covid-19 abates: Wear a mask, follow social distancing recommendations, and avoid crowds.
Listen to the author discuss Singapore’s next stage of containing COVID-19, in the midst of nationwide vaccination:
BOOKMARK THIS: Our comprehensive coverage of the coronavirus outbreak and its developments
Download our app or subscribe to our Telegram channel for the latest updates on the coronavirus outbreak: https://cna.asia/telegram
Professor David Bruce Matchar is Professor of Health Service and Systems Research at the Duke-NUS Medical School.














