commentary Commentary
Commentary: Pharmaceutical firms saved the world with COVID-19 vaccine in record time. But credit goes to others too
Big pharmaceutical companies have played a huge role in rolling out one of the fastest vaccines to be developed in human history. Their highly publicised role belies the other important factors making that impossible task a reality, says Yale-NUS College’s Professor John Driffill.
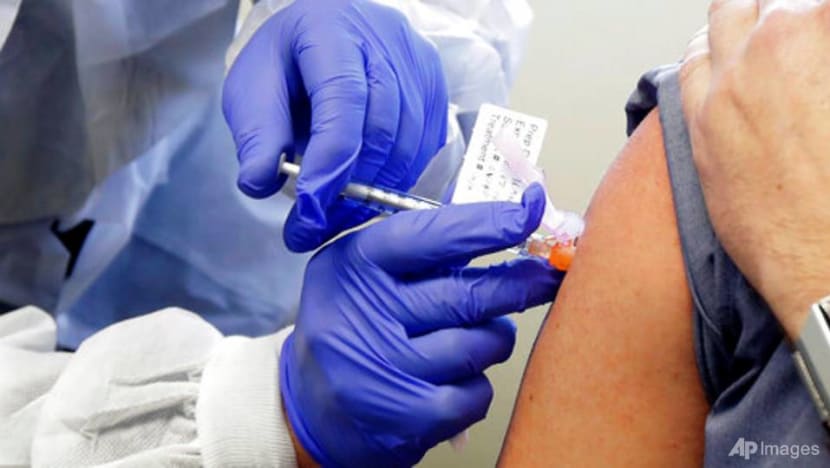
In this March 16, 2020, file photo, Neal Browning receives a shot in the first-stage safety study of a potential vaccine for COVID-19 at the Kaiser Permanente Washington Health Research Institute in Seattle. (Photo: AP/Ted S. Warren)
SINGAPORE: A year since the COVID-19 pandemic hit the world, new vaccines have been approved and are starting to be delivered.
People have begun to be vaccinated in Europe and the United States. In Singapore, healthcare workers were the first to receive the vaccination on Wednesday (Dec 30).
The first vaccine to receive approval was developed by the US multinational pharmaceutical company Pfizer and the German biotechnology firm BioNtech.
This is one of around 200 projects in various countries racing to develop vaccines against COVID-19.
Others on the brink of approval are developed by Moderna in the United States, and AstraZeneca and Oxford University in the UK. Russia has begun to administer its Sputnik vaccine, with at least two approved for emergency use in China, developed by Sinovac and Sinopharm.
Given the scale of the pandemic, the amount of illness, the millions of lives lost, and the economic costs, the financial and reputational rewards to being among the first in the race to develop effective vaccines are enormous.
READ: Commentary: Here’s why taking the vaccine is necessary even if it’s optional
READ: Commentary: A vaccine is on the horizon. But most Singaporeans are adopting a wait-and-see attitude
A BIG TRIUMPH FOR BIG PHARMA
That all this has been achieved in a year is seen as a triumph by large pharmaceutical companies working in partnership with universities and governments.
Such progress generally takes more like 10 years, and the costs of developing a successful new vaccine are usually high, in the region of US$1 billion.
Beyond the research, development and testing, the production and distribution of vaccines on a large scale will be a major undertaking.
There may be demand for 10 billion or more doses in the coming year. The International Air Transport Association (IATA) has estimated that distributing these amounts to 8,000 Boeing-747 cargo-plane loads.
READ: Commentary: Can Singapore be a major COVID-19 vaccine transshipment hub and save its aviation industry?
These rapid developments may give the pharmaceutical industry an opportunity to repair its reputation. The other large corporations involved in distributing vaccines, the airlines and logistics firms, may also enjoy reputational benefits.
Is this a moment when the public perception of such large pharmaceutical multinational corporations gets a boost?
Listen to infectious disease expert outline what's needed to get a vaccine manufactured, transported and administered in our Heart of the Matter podcast:
BIG PHARMA’S POOR REPUTATION
That would be a welcome notion, at least for pharmaceutical companies who have suffered poor reputations in advanced nations for decades.
Award-winning healthcare commentator Ben Goldacre’s 2012 book Bad Pharma detailed a litany of sharp practices by large pharmaceutical companies, influencing research into effects of drugs, suppressing negative results, covertly commissioning research favouring the drugs and lobbying vigorously.
The opioid crisis in the United States is particularly egregious. After a long battle, Purdue Pharma, the makers of the notorious OxyContin drug, has gone into bankruptcy, and the former owners of the company, members of the Sackler family, are being sued for more reparations.
Art galleries and museums that have received donations from them are being pressed to return the funds and remove the Sackler name from their galleries.
Pfizer itself was the subject of a scandal in Nigeria in 1996 over a trial of a meningitis antibiotic, in which several children died. The episode inspired John le Carré’s book The Constant Gardner.
While the industry proclaims that it has since cleaned up its act, doubt remains.
Besides these acts of commission, the industry stands accused of acts of omission. It is not surprising that, as a commercial enterprise, in the pursuit of profits, and despite its enormous resources, the pharmaceutical industry has focused on some types of diseases and treatments and neglected others. Two areas stand out.
First, the world faces a growing threat from antimicrobial resistance, as pathogens develop resistance to existing antibiotics faster than new ones are produced.
We face the prospect that common injuries become life-threatening and much surgery becomes impossible as no treatments can resist hospital-borne bacteria, sending medicine back to the 19th century.
The returns to a private company from developing a new antibiotic do not repay the investment needed, despite the social benefits being vastly greater.

Second, the World Health Organization lists 18 neglected topical diseases (including dengue, rabies, trachoma, sleeping sickness, and river blindness) that continue to afflict populations in many poorer countries.
Big pharma has invested very little in developing treatments for them. Again, the commercial returns do not repay investment.
Happily, in the last few years, partnerships between public bodies, charities, and pharma have been forged to address these omissions.
One hopes these yield positive results. Nonetheless, the laggard role of the industry underlines its pursuit of private gain and neglect of the public good.
READ: Commentary: Making sense of shifting goalposts in public policy and the science of COVID-19
READ: Commentary: What a global COVID-19 exit strategy looks like
A SHOT IN THE ARM FOR PHARMA
Against this background, the success of the pharmaceutical industry, riding to the rescue in the fight against COVID-19, may be a shot in the arm for the public view of big business and a big public relations win.
Only very large corporations have pockets deep enough to invest billions in these risky projects. Only they have the scientific expertise and experience to develop vaccines based on cutting edge technologies and mount the large-scale clinical trials necessary for approval from the regulators.
Nevertheless, the successful development of COVID-19 vaccines has not been the product of big business alone. Rather it has depended on a combination of input from government, universities and other business sectors.
As in the case of Pfizer working with BioNTech, and AstraZeneca with researchers at the University of Oxford, large companies are drawing on the expertise of newer, smaller bio-technology firms and university-funded research.

All these firms employ large numbers of scientists whose advanced education has enjoyed direct and indirect public funding. Big pharmaceutical business draws on a highly skilled labour force whose skills and knowledge have been accumulated by public investment in education.
While the combination of public and private contributions has been essential, market competition has been a factor too.
The payoffs to winning the vaccine development race are likely to be very high (though AstroZeneca has given up some potential profits, offering to sell its vaccine closer to cost to developing countries).
READ: Commentary: Science goes viral, thanks to COVID-19. But there are roadblocks along the way
Governments committed to fast-tracking stringent approval processes and buying billions of doses at an early stage. Some manufacturers have received direct public funding.
There is no doubt Big Pharma has received a reputational boost because of the successes of a few big companies this COVID-19 stricken year. The annual Gallup poll in September testing American attitudes towards the pharmaceutical industry showed almost a 10 percentage point decrease in negative views of the sector.
But the next time you see a rise in the public opinion polls regarding Big Pharma’s work in fighting COVID-19, remember this has been a story of large firms drawing on publicly funded education and research.
They are competing for an enormous market, with governments underpinning demand. They operate in framework of regulations that protects their intellectual property.
The vaccination programme that will ensure everyone who wants a vaccine gets a shot in the arm will be a massive undertaking by national healthcare systems that involve frontline healthcare workers, not to mention requiring the logistical cold chain working at optimal levels to transport billions of vials.
As the prospect of mass vaccination brings an end of the pandemic into view, Big Pharma may be coming to the rescue, but it is not the whole story.
BOOKMARK THIS: Our comprehensive coverage of the coronavirus outbreak and its developments
Download our app or subscribe to our Telegram channel for the latest updates on the coronavirus outbreak: https://cna.asia/telegram
John Driffill is a Visiting Professor of Social Sciences (Economics) at Yale-NUS College. He does research on macroeconomics and monetary policy, institutions and economic performance, and European economies.















