How does the Omicron variant challenge the treatments for COVID-19?
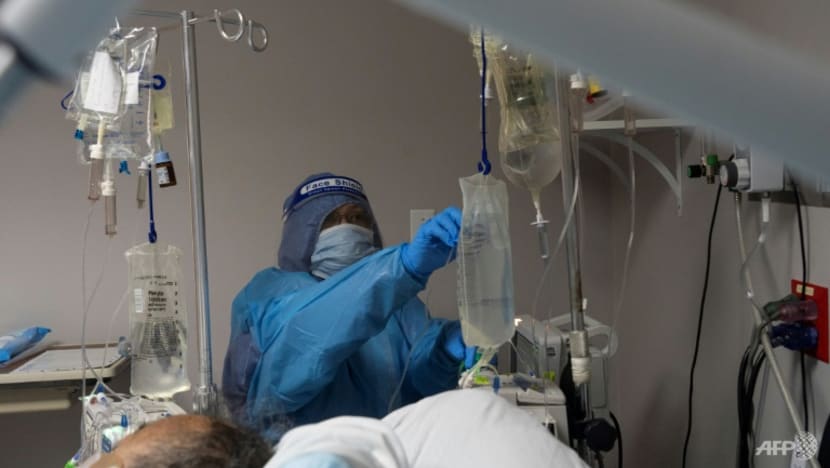
The most effective treatment for COVID-19 currently involves monoclonal antibodies, which are administered via a drip. (File photo: AFP/Getty Images North America/Go Nakamura)
A treatment COVID-19 patients could take by pill to avert life-threatening illness has been something of a holy grail for doctors and drugmakers.
The earliest therapeutics shown to help have typically been administered to patients via a transfusion or once they have become sick enough to require hospitalisation.
Two years after the first COVID-19 cases were reported in China, two pill-based treatments have emerged that even skeptical scientists are hailing as potential game-changers.
Intended for newly infected patients at risk of developing severe disease, the medicines promise to reduce hospitalisations and death as well as make users less likely to spread the coronavirus.
Research is ongoing to determine how well these and other treatments perform against the Omicron variant of the virus identified in late November.
WHAT ARE THE NEW DRUGS?
The first oral treatment, Paxlovid, developed by Pfizer, is a combination of two antiviral pills. One is designed to block the action of a key enzyme that the coronavirus uses to make copies of itself; the other, the HIV medication ritonavir, helps slow the breakdown of the first, enabling it to remain active in the body for longer and at higher concentrations.
The second treatment, molnupiravir, from Merck & Co and Ridgeback Biotherapeutics, was originally developed to treat influenza. The pill inhibits replication of the coronavirus by a mechanism known as lethal mutagenesis.
In simple terms, it causes the machinery that reproduces the virus’ genetic material to make mistakes, thereby rendering the copies defective.
HOW EFFECTIVE ARE THEY?
Paxlovid was shown to reduce the risk of hospitalisation or death by 89 per cent in high-risk COVID-19 patients, according to an interim analysis of a trial reported by Pfizer on Nov 5.
The results were the same whether people started treatment within three or five days of developing symptoms.
Molnupiravir was found to reduce the likelihood of hospitalisation or death by 30 per cent in a study of adults with an increased risk of severe COVID-19, Merck and Ridgeback reported on Nov 26.
HOW DO THESE DRUGS DIFFER FROM OTHER TREATMENTS?
Affordable and easy-to-administer antivirals are ideal treatments, assuming they are safe and well-tolerated, because they directly counter the virus, limiting its damage to the body and the duration of illness.
There are other proven therapeutics that specifically target the coronavirus: Gilead Sciences’ antiviral remdesivir and a number of antibody therapies, which use antibodies produced in a lab to mimic the body’s immune defenses against the virus.
But these are all administered via an intravenous infusion, adding to the complexity and cost of treatment, putting them outside the reach of many developing countries.
And because infusions are usually administered in a hospital or a clinic, infected people risk transmitting the virus to medical staff and other patients.
Other medications have been shown to improve survival in hospitalised patients but do not directly fight the virus; rather they prevent a worsening of COVID-19 symptoms.
These include the cheap steroid dexamethasone, which has been shown to improve survival in COVID-19 patients who need breathing assistance; interleukin-6 receptor blockers, which can suppress a harmful overreaction of the immune system; and blood thinners, used to prevent the blood clots COVID-19 can cause, destroying people’s organs.
HOW WELL WILL TREATMENTS WORK AGAINST THE OMICRON VARIANT?
The World Health Organization (WHO) said in a statement on Nov 28 that steroids and interleukin-6 receptor blockers would still be effective for managing patients with severe COVID-19.
Other treatments are being assessed to see how they perform against Omicron.
Makers of the antiviral pills have said they expect their products to stand up to the new variant. Gilead said the same of its infusion remdesivir.
The antibody therapies are of greater concern because they target the spike protein that the coronavirus uses to enter cells, and Omicron is characterised by 30 or more changes to that region.
Regeneron Pharmaceuticals said on Nov 30 that it was conducting further testing after early evidence suggested that its antibody therapy, REGEN-COV, may be less effective against Omicron.
GlaxoSmithKline expressed confidence on Dec 7 that its antibody treatment, sotrovimab, which it makes in partnership with Vir Biotechnology, would hold up.
WHAT OTHER TYPES OF TREATMENTS ARE BEING INVESTIGATED?
Studies published in September 2020 focused on the role of an immune substance called interferon that helps orchestrate the body’s defenses.
The research found that people with low levels of the substance do poorly in fighting off the coronavirus, suggesting that interferon treatments could help in the early stages of the infection and perhaps prevent life-threatening illness.
However, interferon therapy did not help hospitalised patients in a study reported in October 2021.
A number of COVID-19 treatments have lost favour.
The WHO in December advised against treating COVID-19 patients with convalescent plasma, a basic version of antibody therapy that involves collecting plasma, the liquid part of blood, from people who have recovered from COVID-19 and developed antibodies to the coronavirus, and transfusing it into those who are still sick.
The WHO said the treatment, while costly, does not improve survival or reduce the need for mechanical ventilation.
US regulators in March 2020 authorised the use of the malaria drugs chloroquine and hydroxychloroquine to treat COVID-19, and the next June reversed themselves after determining the drugs were unlikely to work against the coronavirus and could have dangerous side effects.
Related:
HOW DO SCIENTISTS PROVE A TREATMENT IS EFFECTIVE?
Even if a treatment shows promise in laboratory, animal or early human experiments, rigorous testing is needed to prove it is both safe and effective. That requires carefully structured and monitored tests known as clinical trials.
Typically, these studies are designed to show that patients randomly assigned to get the drug do better than those in a control group who do not, and that the results are not a product of chance.
The trials try to subtract all the other influences and factors that might disguise the true effectiveness of the drug.
For one thing, patients often recover from viral illness on their own, or improve because of supportive care such as rest and hydration. And some really sick patients might not respond to treatment no matter how effective it is.
HOW LONG DOES IT TAKE TO PROVE EFFICACY?
It depends. Drugs that are already approved against one infection and shown to be safe can be tested for effectiveness against another in a matter of months.
Experimental drugs may take longer to test, as they have to go through initial studies to evaluate whether they are safe.
Other factors that can slow the process include the supply of drug candidates and the availability of patients to test them in.
The trials also have to be approved by ethics watchdogs and drug regulators. According to a 2017 review, the median time for regulators to approve a new drug in 2015 was 333 days in the US, 422 days in Europe, and 639 days in China. China has since expedited its process.
But when drugs are deemed to fill an immediate need, such as in a pandemic, regulators can speed them through the approval process using a number of paths, including emergency authorisations.
BOOKMARK THIS: Our comprehensive coverage of the COVID-19 pandemic and its developments
Download our app or subscribe to our Telegram channel for the latest updates on the coronavirus pandemic: https://cna.asia/telegram