Singapore authorises Pfizer COVID-19 vaccine for children aged 6 months through 4 years
The three-shot Pfizer vaccine for this age group is expected by the end of the year, while Moderna's will be available next month.
Empty vials of the COVID-19 vaccine of Pfizer-BioNTech. (File photo: AFP/Christof Stache)
SINGAPORE: Pfizer's Comirnaty COVID-19 vaccine has been approved for children aged six months through four years, the Health Sciences Authority (HSA) announced on Thursday (Sep 29).
The vaccination regimen for the primary series in this age group consists of three 3-microgram doses over at least 11 weeks.
The first two doses should be administered three weeks apart, followed by a third dose to be administered at least eight weeks after the second dose.
Official recommendations on the use of this vaccine will be issued by the Expert Committee on COVID-19 Vaccination and the Ministry of Health (MOH), said HSA.
The agency said it had carefully considered the clinical data and assessed that the benefits outweighed the risks for the use of Comirnaty in children aged six months through four years.
"In making this regulatory decision, HSA also consulted experts from its Medicines Advisory Committee and Panel of Infectious Diseases Experts," it added.
This is Singapore's second vaccine authorised for use in young children. The first, Moderna’s two-dose Spikevax COVID-19 vaccine, was in August approved for use in children aged six months up to five years.
The Moderna vaccine in 25 microgram doses will be available next month, while the Pfizer shot is expected to be available by the end of the year, MOH said in response to media queries.
"Vaccinations and boosters remain a critical part of our strategy in ensuring our children are well protected against severe illness, should they be infected with COVID-19," said MOH.
"We encourage everyone who is eligible to be vaccinated when it is offered to them."
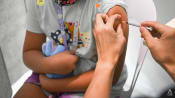
Singapore began vaccinating children aged five to 11 on Dec 27, 2021, after the Comirnaty vaccine was approved for that age group.
Health authorities are preparing to roll out booster shots for this age group in the fourth quarter of the year.
THE CLINICAL DATA
For the Comirnaty vaccine, HSA said the clinical data it reviewed was based on an ongoing Phase 2/3 study conducted by Pfizer, involving about 1,800 participants aged six months through four years.
"The results showed that the immune response in young children with a three-dose primary series was comparable to that in adults aged 16 to 25 years who received two higher doses of 30 micrograms as the primary series vaccine," said HSA.
"Hence, it can be inferred that three doses of the vaccine may provide a similar level of protection in young children as that of two doses in adults."
It added that local real-world data in children aged five to 11 had shown that vaccine effectiveness against the Omicron COVID-19 subvariants was estimated to be around 40 per cent for symptomatic disease.
Vaccine effectiveness also remained high - at more than 80 per cent - for protection against COVID-related hospitalisations for children receiving two doses of Pfizer vaccines.
"It could reasonably be expected that the vaccine would similarly protect younger children aged six months through four years from severe outcomes of COVID-19 such as multisystem inflammatory syndrome and other potential complications," said HSA.
Preliminary data from a secondary analysis of COVID-19 cases in the clinical study also suggested that vaccine efficacy was estimated to be around 73 per cent.
But "the evidence was limited and should be interpreted with caution", HSA warned.
ADVERSE EVENTS
The clinical studies also revealed that adverse events in young children were similar to those reported in adults.
These were mild to moderate in severity and commonly reported with childhood vaccination, such as injection site pain, fever, fatigue and headache, said HSA.
"These reactions are generally associated with vaccinations and expected as part of the body’s natural response to build immunity against COVID-19. They usually resolve on their own within a few days," added the authority.
HSA said there were no cases of serious adverse events such as myocarditis or pericarditis (inflammation of the heart muscle) reported in the clinical study with Comirnaty.
But caregivers of young children should monitor for signs and symptoms of myocarditis such as chest pain and breathing difficulty, and also minimise rigorous physical activity following vaccination, said HSA.
The authority said it would continue to actively monitor the safety of the vaccine and require Pfizer to submit data from the ongoing clinical study.
"HSA will take the necessary actions and provide updates to the public if any significant safety concerns are identified," it added.
BOOKMARK THIS: Our comprehensive coverage of the COVID-19 pandemic and its developments
Download our app or subscribe to our Telegram channel for the latest updates on the coronavirus pandemic: https://cna.asia/telegram