'Potent' medicinal ingredients, banned substance found in five health products

(From left) X-Gout, dcr Natural Herbs Honey Enzyme, KMS2 Dark Chocolate Mocha Botanical Beverage, Speedy Slim Capsules (Black) and Speedy Slim Capsules (Gold).
SINGAPORE: The Health Sciences Authority (HSA) raised an alert on Thursday (Dec 30) after tests found "potent adulterants", including medicinal ingredients and a banned substance, in five health products.
These adulterants, which were sold on local e-commerce and social media platforms, had caused serious adverse effects in consumers, said HSA in a news release.
Members of the public are advised not to purchase or consume the following products - X-Gout, dcr Natural Herbs Honey Enzyme, KMS2 Dark Chocolate Mocha Botanical Beverage, Speedy Slim Capsules (Black) and Speedy Slim Capsules (Gold).
"HSA has worked with the platform administrators to promptly remove the affected listings and issued warnings to the respective sellers," said the authority.
X-GOUT

After taking X-Gout for a year, a woman in her 40s developed Cushing’s syndrome, a life-threatening steroid-induced condition.
Her friend had purchased the product via Shopee Malaysia on her behalf and encouraged her to use it for her knee pain.
The woman gained 22kg over a few months, experienced shortness of breath and swelling of a lower limb. She was diagnosed with diabetes when she consulted her doctor on the symptoms.
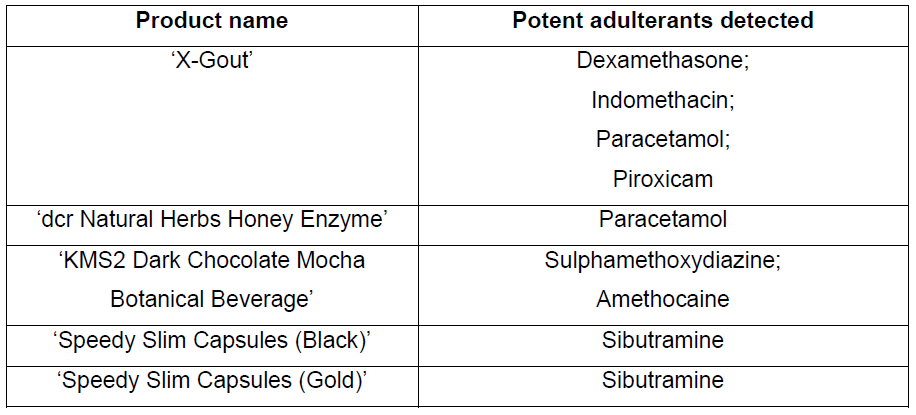
As these symptoms indicated inappropriate steroid consumption, her doctor reported the case to HSA for investigation. HSA tested the product and detected four medicinal ingredients: dexamethasone, a steroid; indomethacin and piroxicam, which are both non-steroidal anti-inflammatory drugs (NSAID), and paracetamol.
Long-term unsupervised use of steroids such as dexamethasone can cause increased blood glucose levels (which may lead to diabetes), Cushing’s syndrome (characterised by a round face or "moon face" appearance and upper body obesity with thin limbs) and other serious adverse effects.
Consumers who have taken X-Gout should see a doctor as soon as possible, said HSA.
Discontinuation of steroids without proper medical supervision can also cause serious withdrawal symptoms such as fatigue, confusion and low blood pressure, especially when the product has been taken for more than a few weeks.
DCR NATURAL HERBS HONEY ENZYME

A man in his 40s developed Cushing’s syndrome after taking dcr Natural Herbs Honey Enzyme for about 6 months.
When he stopped using the product, he experienced withdrawal symptoms such as loss of appetite, lower energy levels and rashes on his body. Based on the man’s symptoms, his doctor suspected that the product was adulterated with steroids and alerted HSA.
HSA obtained samples of the product which were tested and found to contain paracetamol.
"It cannot be ruled out that there may potentially be other adulterants or steroids present in other batches of the product, since Cushing’s syndrome is often associated with long-term inappropriate consumption of steroids," said the authority.
Those who have taken dcr Natural Herbs Honey Enzyme should see a doctor as soon as possible, as there may potentially be potent ingredients in other batches of the product that have not been detected.
KMS2 DARK CHOCOLATE MOCHA BOTANICAL BEVERAGE

HSA received feedback from a member of the public who had experienced side effects such as fast heartbeat, thirst, and dry mouth after consuming KMS2 Dark Chocolate Mocha Botanical Beverage for a few days.
The sellers had made claims that the product “speeds up fat burning by inducing heat, helps soothe digestive systems and boost metabolic cycles to reduce appetite and is safe without rebounding weight”.
HSA tested the product and found them to contain sulphamethoxydiazine, an antibacterial medicine, and amethocaine, an anaesthetic.
Consumers should stop taking the product immediately and consult a doctor if they feel unwell or are concerned about their health.
SPEEDY SLIM CAPSULES

HSA detected and seized Speedy Slim Capsules (Black) and Speedy Slim Capsules (Gold) following feedback from members of the public that there were unapproved slimming products being sold on Instagram.
These products made exaggerated claims such as “target at breaking down stubborn extra fat”, “cut off starch absorption”, and “losing weight and clearing fat”.
They were subsequently found to contain very high levels of sibutramine, a prescription-only weight loss medicine that has been banned since 2010 due to an increased risk of heart attack and stroke.
Other serious adverse effects reported in the past by consumers who took products adulterated with sibutramine include insomnia and hallucinations.
In 2019, a consumer experienced extremely fast heart rate and became unconscious. She was resuscitated and suffered debilitating consequences, including requiring the implantation of a defibrillator (a medical device) to help her heart to function.
Those who have taken Speedy Slim Capsules are advised to stop consuming them immediately and consult a doctor if they feel unwell or are concerned about their health.
"There is no quick and easy way to lose weight," said HSA.
"Weight control should be achieved through a combination of balanced diet and appropriate exercise. Those who need help managing their weight should consult a doctor, dietitian or a healthcare professional."
ADVISORY TO SELLERS AND SUPPLIERS
It added that consumers should be wary of products that carry exaggerated claims or deliver unexpectedly quick results, such as fast weight loss or immediate pain relief. This is because they can contain potent ingredients that can seriously harm your health.
Consumers should also exercise caution when buying such products online or from well-meaning friends as they cannot be certain where and how these products were made and whether they have been adulterated with harmful ingredients.
All sellers and suppliers must stop selling these products immediately, added HSA.
The authority said it will not hesitate to take stern enforcement actions against anyone who sells and/or supplies products found to be adulterated with potent medicinal ingredients.
Sellers and suppliers can be prosecuted. If convicted, they can be jailed for up to 3 years and/or fined up to S$100,000.














