Undeclared ingredients found in three health products, HSA warns


SINGAPORE — A man in his forties bought a health product overseas through a friend and ended up in hospital for Cushing’s syndrome, joint pain, weight gain and insomnia.
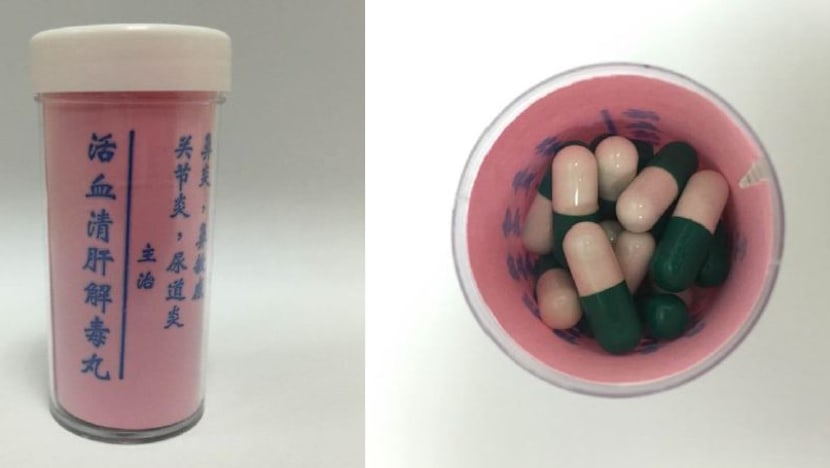
He had consumed a product called ‘Huo Xue Qing Gan Jie Du Wan’, which contains dexamethasone – a potent steroid – and could have contributed to his medical condition.
It is one of three Chinese health products that have been found to contain undeclared western medicinal ingredients, and the Health Sciences Authority (HSA) is warning the public not to buy or consume them.
All three products were not properly labelled, but were tested to contain ingredients that can cause serious adverse reactions, HSA said in a media release today.
The other two products are Huang Niu Mu Shi Ni De Jiu Xing and Meizitang Botanical Slimming 100% Natural Soft Gel.
Both Huo Xue Qing Gan Jie Du Wan and Huang Niu Mu Shi Ni De Jiu Xing were purchased overseas and claimed to treat various forms of chronic ailments such as arthritis, liver disorders, high blood pressure, diabetes and stroke.
Both were found to contain the potent steroid dexamethasone, which can also cause high blood pressure, cataracts, and increased risk of infections, among others.
Another ingredient found in both products was Chlorpheniramine - an antihistamine used to relieve allergic reactions such as rhinitis - and which can only be used under medical supervision by a pharmacist or doctor. Some of the side effects of Chlorpheniramine include drowsiness, blurred vision, vomiting, constipation and poor coordination.
HSA has warned, however, that consumers who are already consuming these products not to make a sudden stop as an abrupt discontinuation may cause serious withdrawal symptoms. Instead, they should consult a doctor as soon as possible. The ‘Meizitang Botanical Slimming 100% Natural Soft Gel, was sold via online platforms and claimed to be ‘free of side effects’. But, it was tested to contain diclofenac, a potent painkiller that may potentially cause serious gastric bleeding, heart attacks and stroke, especially if used for a prolonged period. “It should be used under close medical supervision, especially in patients with underlying heart conditions,” HSA advised.
Those caught selling any of these three adulterated health products may face a fine of up to S$10,000 and/or jailed for up to two years under the Poisons Act. HSA also noted that it is the seller’s responsibility to ensure that the
products they sell are safe and adhere to local legislations before they are made available in the market.












